|
The signs of the ions are only used to determine the relative order in which the ion symbols are written.In this example, Be 2 will be written before CN – 1 in the final chemical formula. In order to ensure consistent formatting in all ionic chemical formulas, the symbol for the cation is written first.Based on the information presented in the previous sections of this chapter, the cyanide ion is symbolized as CN – 1, and beryllium ionizes to form Be 2. Write the ion symbols for any indicated polyatomic ions and for any monatomic ion that results upon the ionization of a main group or transition metal element.After establishing that a pair of chemicals will form an ionic bond, a five-step process can be employed to determine the chemical formula of the resultant ionic compound. In the given combination, the cyanide ion is classified as a polyatomic anion, and beryllium, a metal, ionizes to form a cation. Recall that an ionic bond is produced when a cation exists in close physical proximity to an anion, creating an electrostatic attractive force. The procedure for determining the chemical formula of an ionic compound containing exclusively main group elements or a combination of main group and transition metal elements can also be utilized to establish the chemical formula of an ionic compound that contains a polyatomic ion.įor example, consider the cyanide ion and beryllium.īased on the combinations listed in Section 3.2, the cyanide ion and beryllium will combine to form an ionic compound. Writing Chemical Formulas of Ionic Compounds Containing Polyatomic Ions In the purification of drinking water and wastewater treatment plants, it encourages particle collision by neutralising charge.\) Aluminium Sulfate is used as a coagulant.The liquid is then evaporated and allowed to crystallise. Aluminium sulfate is created by reacting freshly precipitated aluminium hydroxide with the appropriate amount of sulphuric acid.Its formula is XAl(SO 4) 2.12H 2O, where X is a monovalent cation such as potassium or ammonium. In general, the term "alum" is used to refer to any double sulphate salt. 
In some industries, aluminium sulfate is referred to as alum or papermaker's alum.For example, it is found in volcanic areas and on coal-mining waste dumps that are burning. Millosevichite, a rare mineral, contains the anhydrous form of Aluminium Sulfate. 
Both types are non-combustible and non-toxic. Aluminium sulfate is a colourless liquid in its solution form and a white crystalline solid in its anhydrous form.Water purification employs the use of aluminium sulphate.Aluminium sulfate is used in gardening to help balance the pH of the soil.It is employed in the dyeing of clothing.Aluminium sulfate is utilised often as a fireproofing agent.It is also utilised in water treatment plants for sewage treatment.Aluminium sulfate is utilised in the production of firefighting foam.It is used as a waterproofing agent and an accelerator in concrete.It's also used to create a variety of prints on fabric.Aluminium sulfate is used in the production of paper.It is then decanted from any sediment after being drawn off and evaporated until it reaches a specific gravity of 1.40. This solution is left to sit for a while before being evaporated till the iron (II) sulphate crystallises on cooling. After that, it is extracted using water and an Aluminium Sulfate solution with a specific gravity of 1.16. It reacts with the clay to make Aluminium Sulfate, which is similar to what happens when the air weathers. 
Sulfuric acid is produced during the roasting process. These are either roasted or exposed to the air. Upper Bavaria, Bohemia, Belgium, and Scotland are all home to these. Iron pyrite, aluminium silicate, and numerous bituminous compounds make up alum. 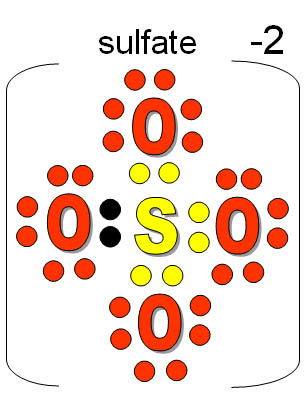
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
